Eagle Medical provides industry-leading sterilization services for medical devices using multiple validated methods. Our state-of-the-art facilities offer Ethylene Oxide (EtO), Hydrogen Peroxide Gas Plasma (HPGP), Gamma radiation, and Electron Beam (E-beam) sterilization.
FDA-compliant processes meeting ISO standards
Multiple sterilization modalities for different device requirements
Validation and documentation support for regulatory submissions
Temperature-sensitive options for electronics and delicate materials
Fast turnaround times to accelerate time-to-market
All sterilization processes are performed in ISO 13485:2016 certified facilities with comprehensive quality systems and full traceability documentation.

An FDA Established Category A form of sterilization, compliant with ISO 22441:2022 standards, that provides low-temperature, electronics (NVRAM) and battery friendly sterilization process for your complex software driven electro-medical devices.
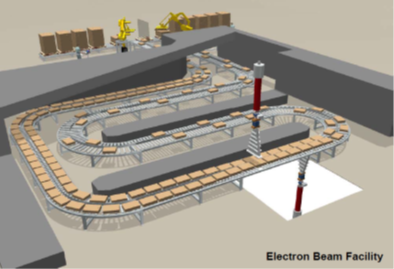
Uses high energy electrons to penetrate low-density packaging and sterilize products quickly and effectively. This radiation-based sterilization method offers rapid processing with excellent material compatibility.

With high penetrating power, Gamma Beam Irradiation can sterilize the most dense products and packages when compared with E-Beam Sterilization. This deep-penetrating radiation method handles complex, dense packaging configurations.
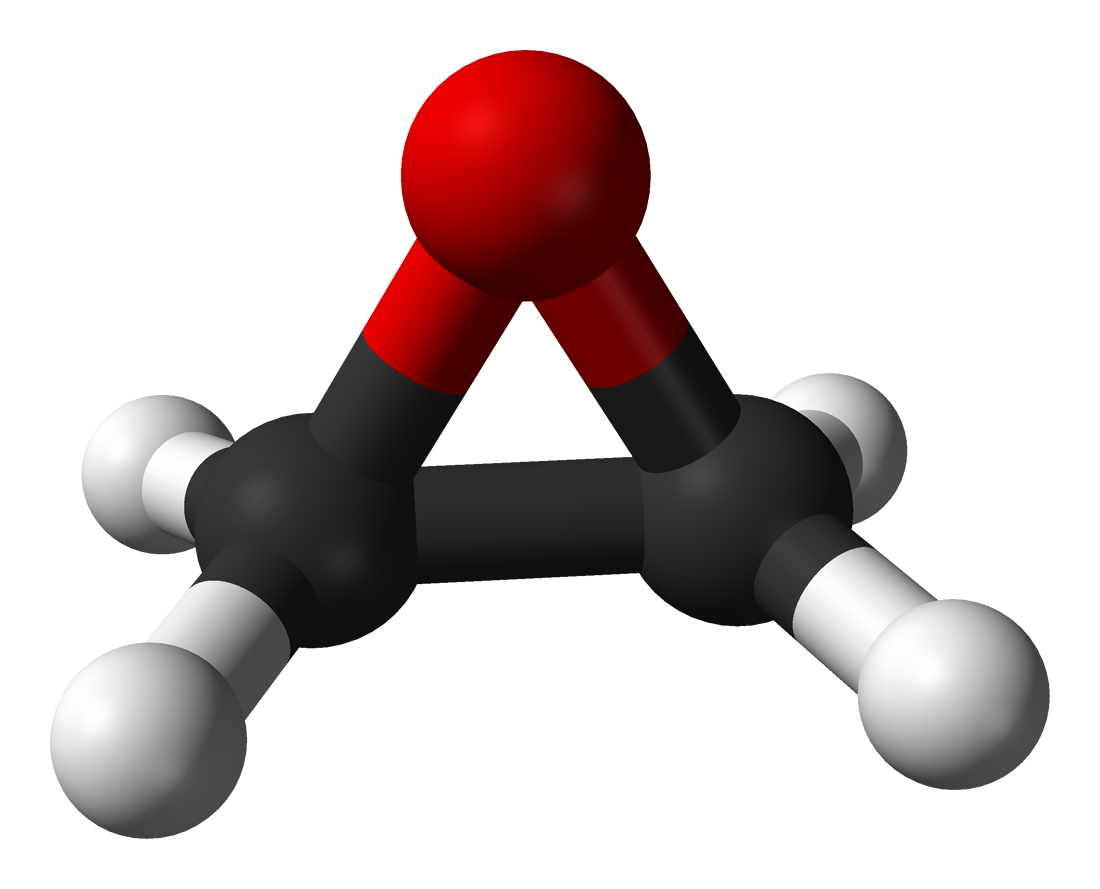
One of the most widely used sterilization methods, Ethylene Oxide is a versatile sterilization solution that can penetrate thin films and breathable materials such as Tyvek. This chemical sterilization method offers excellent material compatibility.
Contact our experts to discuss your specific medical device needs and find the right solution for your project.